Histopathological Evaluation of Intestinal Eimeria and Ogmocotyle-like Trematode Infections in Slaughtered Dromedary Camels
DOI:
https://doi.org/10.71336/ijvar.720Keywords:
Coccidiosis, Parasitic Diseases, Protozoan Infections, Small Intestine, TrematodaAbstract
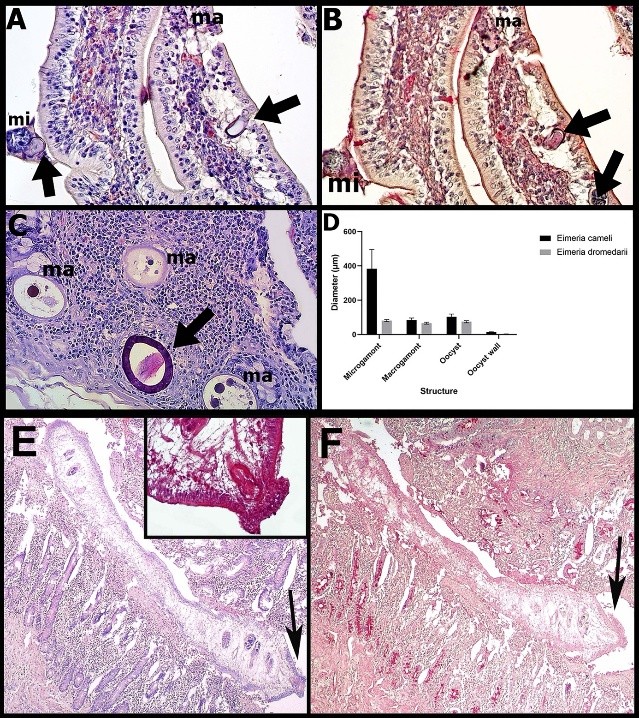
Camelus dromedarius is a resilient, desert-adapted species; however, it remains susceptible to gastrointestinal parasitic infections that often go undiagnosed in clinically healthy individuals. This study investigated the occurrence, anatomical distribution, and histopathological impact of protozoan and helminth parasites in the small intestinal segments (duodenum, jejunum, and ileum) of 14 asymptomatic dromedaries slaughtered in Blida, Algeria. Histopathological analyses revealed subacute catarrhal enteritis, villous atrophy, epithelial degeneration, and inflammatory infiltration. Sexual stages of Eimeria cameli and Eimeria dromedarii were frequently identified, particularly within the crypts of Lieberkühn and villous epithelium, respectively. Overall, 78.57% of animals were found to be parasitized, with E. cameli present in 57.14%, and one individual harboring both species. Morphometric and Periodic Acid–Schiff (PAS) staining supported the identification of distinct developmental stages of both species. Notably, Ogmocotyle-like trematodes were detected in the duodenum of two camels, representing the first report of such organisms in this host. These flukes were associated with focal epithelial necrosis, eosinophilic infiltration, and luminal debris, underscoring their pathological significance. Despite the absence of molecular confirmation, histopathology, histochemistry, and morphometric analysis collectively enabled a detailed characterization of these infections. These findings highlight the importance of post-mortem surveillance and accurate parasitic identification in camels. Future studies should incorporate molecular tools to enhance species-level resolution and explore host–parasite immunological dynamics.
References
Bandyopadhyay S, Pal P, Bhattacharya D, Bera AK, Pan D, Rahman H. 2010. A report on the prevalence of gastrointestinal parasites in yaks (Bos poephagus) in the cold desert area of North Sikkim, India. Tropical Animal Health and Production, 42: 119–121. https://doi.org/10.1007/s11250-009-9394-8 DOI: https://doi.org/10.1007/s11250-009-9394-8
Bouragba M, Laatamna A, Cheddad FE, Baroudi D, Houali K, Hakem A. 2020. Gastrointestinal parasites of dromedary camel (Camelus dromedarius) in Algeria. Veterinary World, 13(8):1635–1640. https://doi.org/10.14202/vetworld.2020.1635-1640 DOI: https://doi.org/10.14202/vetworld.2020.1635-1640
Choe SJ, Na KJ, Jee CH. 2011. A survey on gastrointestinal parasites of Korean water deer (Hydropotes inermis argyropus) in Chungbuk Province. Korean Journal of Veterinary Service, 34: 259–262. https://doi.org/10.7853/kjvs.2011.34.3.259 DOI: https://doi.org/10.7853/kjvs.2011.34.3.259
Dubey JP, Schuster RK. 2018. A review of coccidiosis in Old World camels. Veterinary Parasitology, 262: 75–83. https://doi.org/10.1016/j.vetpar.2018.08.008 DOI: https://doi.org/10.1016/j.vetpar.2018.08.008
Dubey JP. 2019. Coccidiosis in Livestock, Poultry, Companion Animals, and Humans, In: Dubey JP, Schuster RK. (Eds.), Coccidiosis in Old World Camels, 1st Ed. Routledge: CRC Press, Newyork, US, pp. 147-152. https://doi.org/10.1201/9780429294105-12 DOI: https://doi.org/10.1201/9780429294105-12
Dubey JP. 2018. A review of coccidiosis in South American camelids. Parasitology Research, 117:1999–2013. https://doi.org/10.1007/s00436-018-5890-y DOI: https://doi.org/10.1007/s00436-018-5890-y
El-Khabaz KAS, Abdel-Hakeem SS, Arfa MI. 2019. Protozoan and helminth parasites endorsed by imported camels (Camelus dromedarius) to Egypt. Journal of Parasitic Diseases, 43:607–615. https://doi.org/10.1007/s12639-019-01138-y DOI: https://doi.org/10.1007/s12639-019-01138-y
Faye B, Konuspayeva G. 2012. The encounter between Bactrian and Dromedary camels in Central Asia. In: Knoll M, Burger P. (Eds.), Camels in Asia and North Africa: Interdisciplinary perspectives on their past and present significance, 1st Ed. Academy of Sciences Press, Wienne, Austrian, pp. 27-33.
Hussein HS, Kasim AA, Shawa YR. 1987. The prevalence and pathology of Eimeria infections in camels in Saudi Arabia. Journal of Comparative Pathology, 97(3): 293–297. https://doi.org/10.1016/0021-9975(87)90093-4 DOI: https://doi.org/10.1016/0021-9975(87)90093-4
Iwaki T, Okada T, Seki K, Izawa K, Sakurai F. 2012. Ogmocotyle ailuri (Price, 1954) (Digenea: Notocotylidae) found in the Japanese monkey, Macaca fuscata. Journal of Veterinary Medical Science, 74: 1211–1212. https://doi.org/10.1292/jvms.11-0572 DOI: https://doi.org/10.1292/jvms.11-0572
Junker K, Horak IG, Penzhorn B. 2015. History and development of research on wildlife parasites in southern Africa, with emphasis on terrestrial mammals, especially ungulates. International Journal for Parasitology: Parasites and Wildlife, 4: 50–70. https://doi.org/10.1016/j.ijppaw.2014.12.003 DOI: https://doi.org/10.1016/j.ijppaw.2014.12.003
Kheirandish R, Nourollahi-Fard SR, Faryabi Z. 2012. Prevalence and pathologic study of Eimeria cameli in slaughtered camels. Eurasian Journal of Veterinary Science, 28(3): 138–141.
Luna LG. 1968. Routine Harris hematoxylin and eosin stain. Histopathology Laboratories, Armed Forces Institute of Pathology, 3rd Ed. Washington DC, US, pp. 38-39.
Mohandesan E, Fitak RR, Corander J, Yadamsuren A, Chuluunbat B, Abdelhadi O, Raziq A, Nagy P, Stalder G, Walzer C, Faye B & Burger PA. 2017. Mitogenome sequencing in the genus Camelus reveals evidence for purifying selection and long-term divergence between wild and domestic Bactrian camels. Scientific reports, 7(1): 9978. DOI: https://doi.org/10.1038/s41598-017-08995-8
Parsani HR, Singh V, Momin RR. 2008. Common parasitic diseases of camel. Veterinary World, 1(10): 317–318.
Sazmand A, Joachim A. 2017. Parasitic diseases of camels in Iran (1931–2017) - a literature review. Parasite, 24: 1-15. https://doi.org/10.1051/parasite/2017024 DOI: https://doi.org/10.1051/parasite/2017024
Zhao Y, Zhang K, Zou M, Sun Y, Peng X. 2018. gga-miR-451 negatively regulates Mycoplasma gallisepticum (HS strain)-induced inflammatory cytokine production via targeting YWHAZ. International Journal of Molecular Sciences, 19(4):1191. https://doi.org/10.3390/ijms19041191 DOI: https://doi.org/10.3390/ijms19041191
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 International Journal of Veterinary and Animal Research (IJVAR)

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.










